Design & Technology students often learn about ferrous metals, non-ferrous metals and their alloys. This article summarises the properties, stock forms, and uses of common metals, helping students to revise these topics.

Metals are separated into two categories: ferrous and non-ferrous.
Ferrous Metals
- Contain iron (“ferrous” comes from the Latin word “ferrum,” meaning iron – this is why the symbol of iron is Fe)
- Magnetic (although some alloys, like stainless steel are not always magnetic)
- Generally have high tensile strength (hence valuable for construction and manufacturing)
- Prone to rust when exposed to oxygen and moisture due to iron content
- Become less malleable as more carbon is added
An alloy is a mixture of two or more elements, where at least one of these is a metal. Many alloys are mixtures of 2 or more metals. These are combined to improve appearance or properties.
FERROUS METALS & THEIR ALLOYS
Cast iron
Properties
Uses
Wrought iron is different (has less carbon) and is hammered/forged into shape
Mild steel
Properties
Uses
As the carbon content in increases, steel becomes harder and stronger but more difficult to work with.
High-carbon steel
Properties
Uses
Medium carbon steel is useful for things like a woodworking vise etc
High-speed steel
Properties
Uses
Stainless steel
Properties
Uses
Non-ferrous metals
- Do not contain iron
- Not magnetic
- Generally more corrosion resistant than ferrous metals
- Often more expensive
NON-FERROUS METALS & THEIR ALLOYS
Aluminium
Properties
Uses
Malleable – hammered and pressed into shape without breaking
Ductile – stretches into wire without breaking
Copper
Properties
Uses
Copper is a self-finishing material – obtains a green patina with age as oxidises upon exposure to air / moisture
Lead
Properties
Uses
Tin
Properties
Uses
Rarely used in pure form as it is very soft
Zinc
Properties
Uses
Titanium
Properties
Uses
Nickel-titanium (an alloy made from from nickel and titanium) is the most common shape memory alloy
Tungsten
Properties
Uses
Duralumin
Properties
Uses
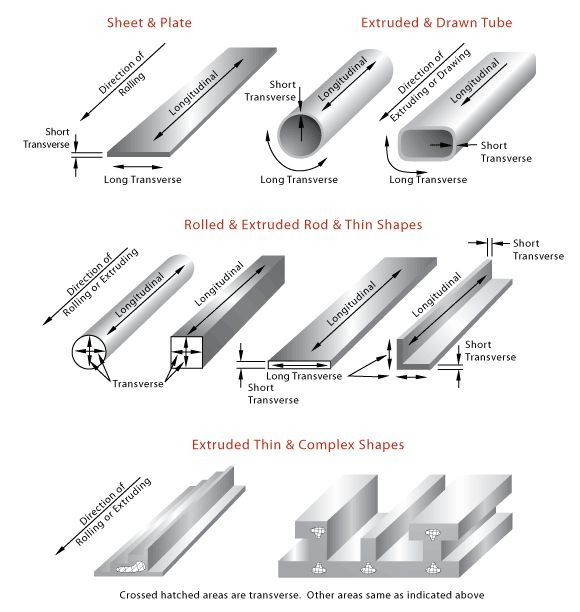
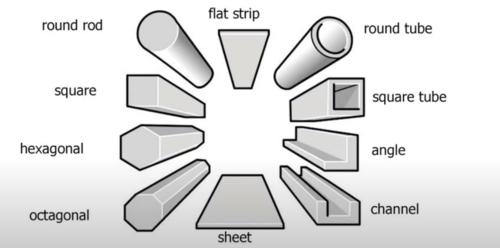
Common stock forms of metals
Environmental Considerations
Learn about other modern materials that contain metals (including metal foams and super alloys), as well as smart materials (including shape-memory alloys).
Metal enhancement (A2 students only)
These methods change the internal structure and properties of a metal through controlled heating and cooling.
- Case hardening: a heat treatment process used to produce a hard, wear-resistant outer layer (the “case”) while leaving the inner core soft, tough, and able to absorb shock. Used for low-carbon steels such as mild steel. Typically carried out using a brazing hearth and torch. The steel component is heated until cherry red, then plunged into carbon-rich case-hardening compound (a powder). The heat causes carbon from the compound to merge with the outer layer of the steel. The component is then reheated and cooled immediately in cold, clean water. This rapid cooling forms a hard surface, while the low-carbon core remains unaffected and retains its toughness. The cycle of heating, coating, and quenching can be repeated to build up a thicker hardened case. The result combines two useful properties that would otherwise be difficult to achieve in a single piece of metal: a hard surface that resists wear, scratching, and abrasion, and a tough core that resists cracking or shattering under impact. This makes case-hardening particularly suitable for tools and mechanical parts, where the working edges need to stay sharp and resist wear during repeated use. Safety precautions: wear eye protection (goggles or a face shield), tie back long hair, keep fingers well clear of the hot metal, using tongs to handle the workpiece, wear heat-resistant gloves and leather apron. Work in a well-ventilated area, as case-hardening compounds can give off fumes, and be aware of the fire risk associated with the brazing hearth.
- Annealing: When you bend, hammer and work metal it becomes increasingly stressed and brittle. For example, if you bend a piece of wire back and forward it will quite quickly snap in two. Annealing is a heating process that reverses this, restoring the metal to its original quality, softening the metail and relieving internal stresses. To do this, the metal is heated to a specific temperature (which varies by material), held there long enough for the internal structure to reorganise, and then cooled very slowly — usually by leaving it in the furnace or burying it in sand or ash. This slow cooling allows the metal to become uniform and stress-free. Annealing is often used on steel, copper, and aluminium after they have been work-hardened through bending, hammering, or rolling, restoring their ductility so they can be worked further without cracking.
- Normalising: A heat treatment process applied to steel to produce a more uniform, slightly tougher material than annealing. The steel is heated to just above its critical temperature (similar to annealing) but is then cooled in still air rather than in the furnace. This faster cooling results in a metal that is stronger and tougher than annealing but still reasonably easy to machine. Like annealing, normalising is often used to remove internal stresses caused by forging, welding, or casting, and to prepare steel for further heat treatment.
- Hardening: A heat treatment process used to make medium- and high-carbon steels hard and wear-resistant. The steel is heated to its critical temperature (cherry red) and then quenched rapidly in water, oil, or brine (salty water). This sudden cooling traps the internal structure in a hard but brittle state. The result is a metal that is very hard and resists wear, but is also brittle and prone to cracking or shattering under impact — which is why hardening is almost always followed by tempering (see below). Note that hardening only works on steels with enough carbon content; low-carbon steels like mild steel must be case hardened instead (which adds sufficient carbon to the exterior).
- Tempering: A heat treatment process carried out after hardening to reduce brittleness and restore some toughness to the metal. The hardened steel is cleaned and polished, then reheated gently to a relatively low temperature (typically between 200°C and 300°C) and held there before being cooled in liquid again. As the steel heats, its surface changes colour in a predictable sequence — pale straw, dark straw, brown, purple, blue — and the colour reached indicates the final properties. Pale straw is used for tools that need to stay very hard, such as scribers and lathe tools, while darker colours like blue are used for springs and tools that need to flex without snapping. Tempering gives the final balance of hardness and toughness appropriate to the tool’s purpose.
AS Design & Technology examination questions
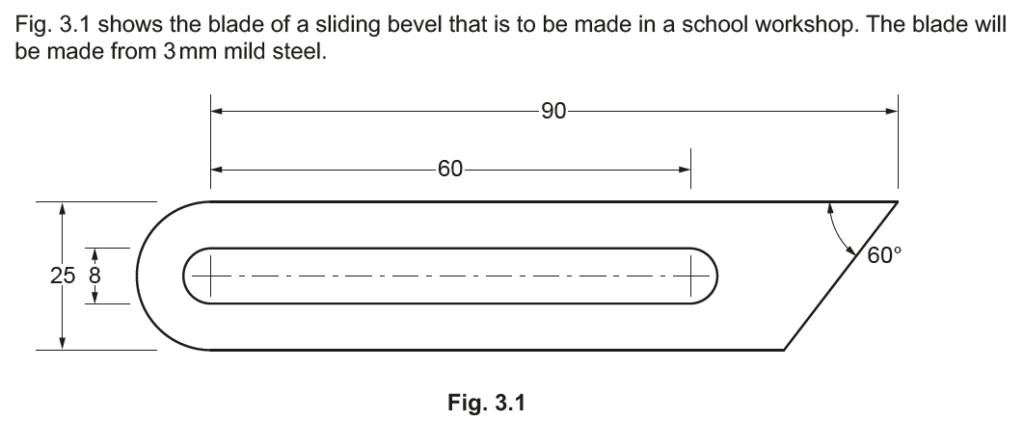



Examiner comment: This question was generally well attempted, with ease of machining and cost effective often given.



Examiner comment: Most candidates were able to state at least one reason with aesthetic appeal and corrosion resistant being the most popular answers.
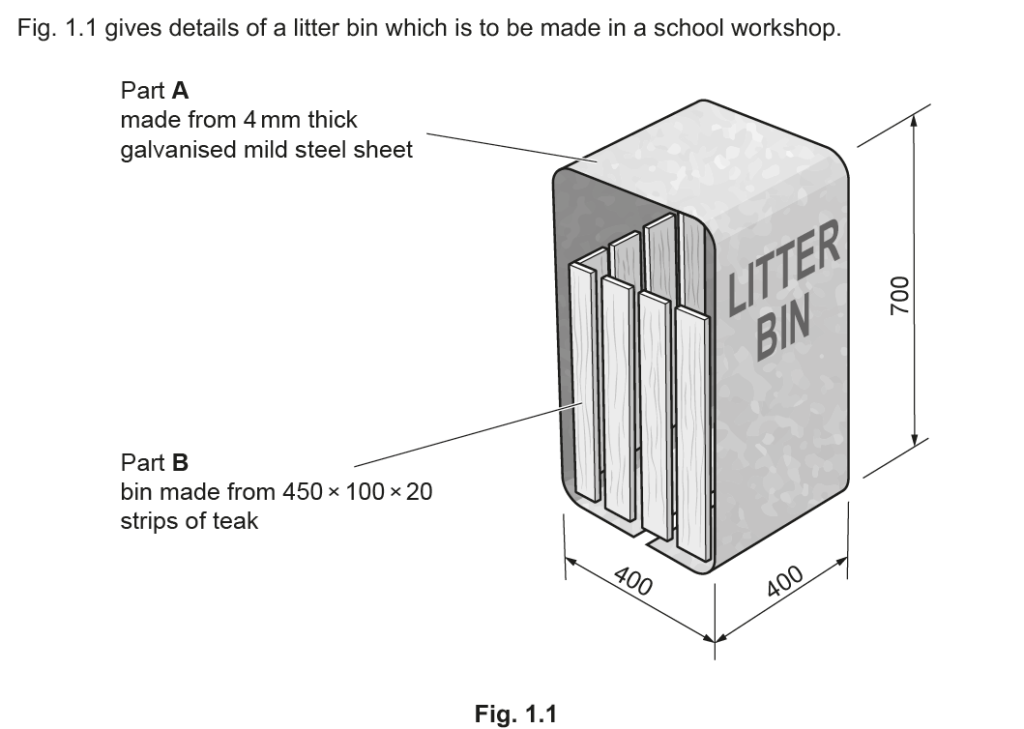


Examiner comment: This was generally answered well and most candidates achieved one mark, and many gained both marks with ‘will not rust’ given regularly as a correct answer.
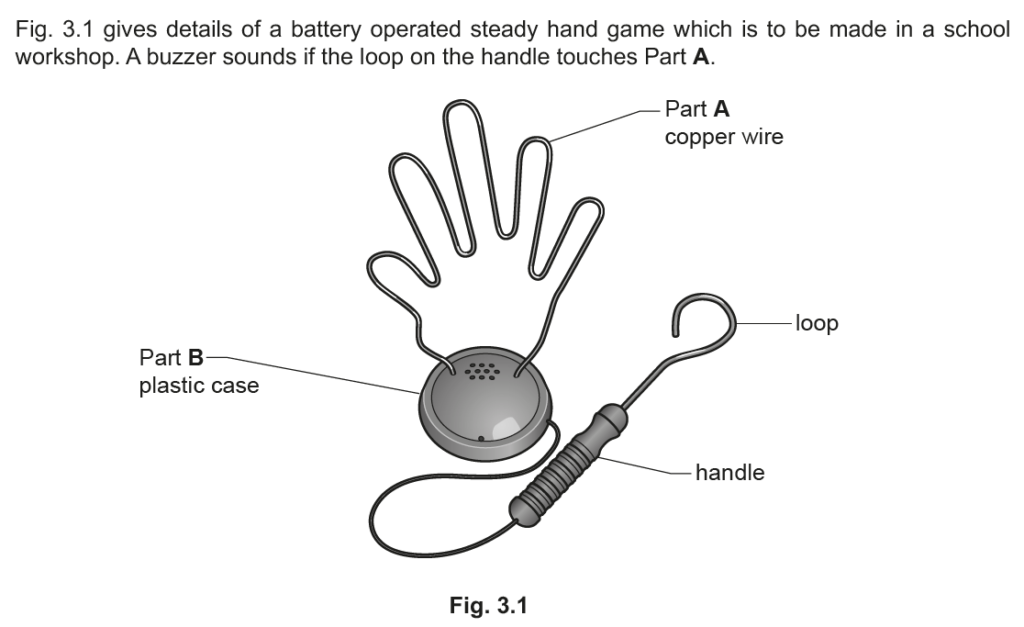


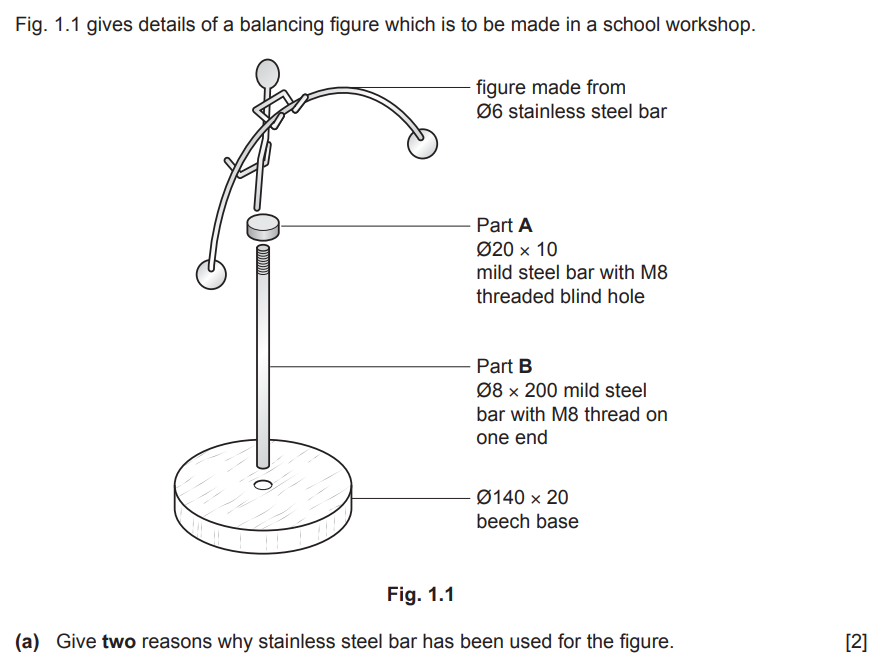

Examiner comment: Generally, this was answered well and most candidates could give two reasons why stainless steel had been used for the figure.
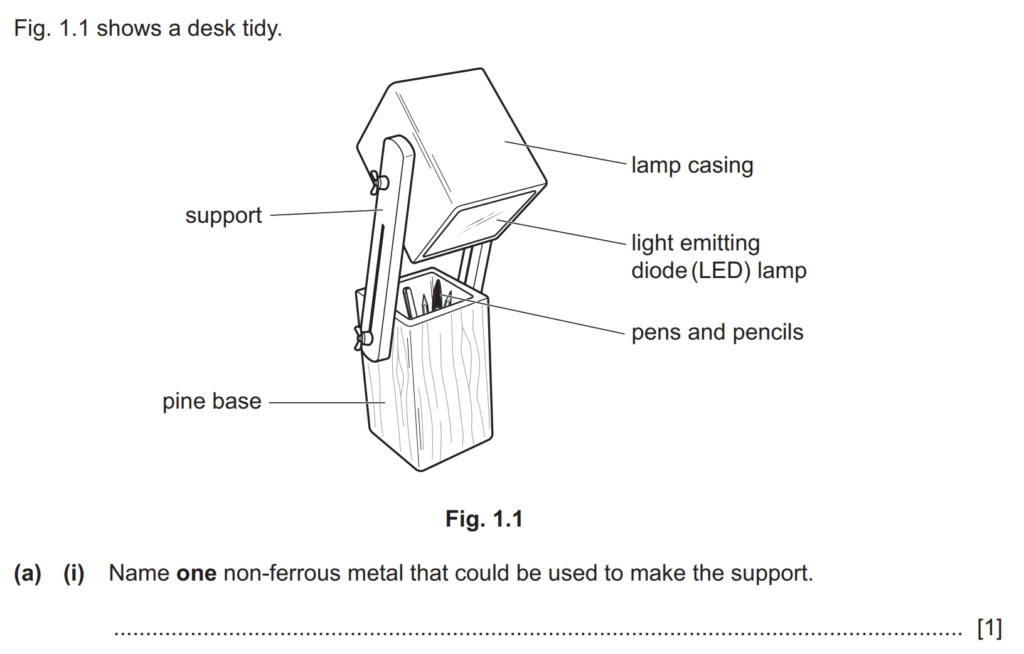

Examiner comment: Most candidates were able to name a non-ferrous metal that could be used to make the support for the desk tidy. Frequently seen correct answers included aluminium, brass and copper. Candidates who gave an incorrect answer often named a ferrous metal, such as iron.
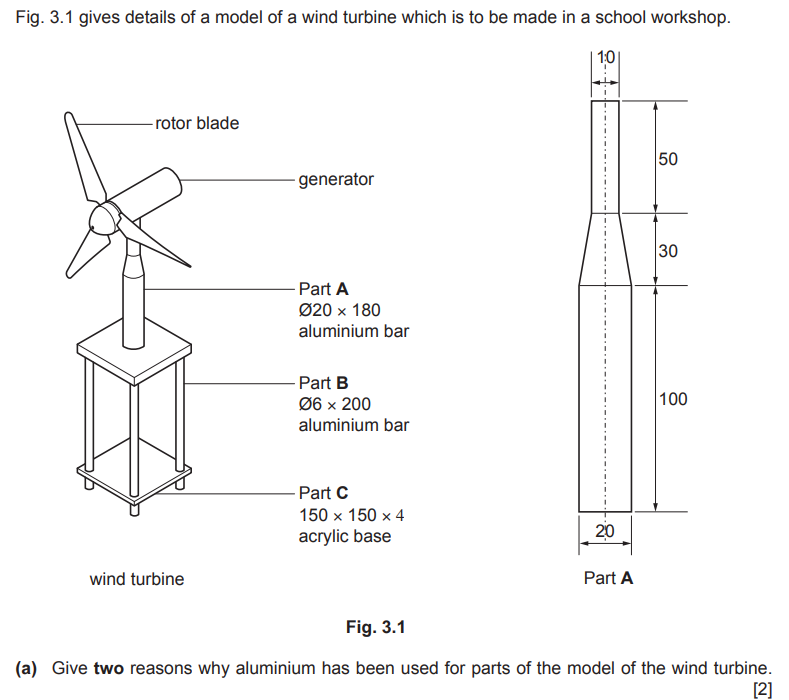

Examiner comment: Most candidates gave at least one reason for using aluminium for the parts of the model wind turbine, with ‘lightweight’ being a very popular answer.
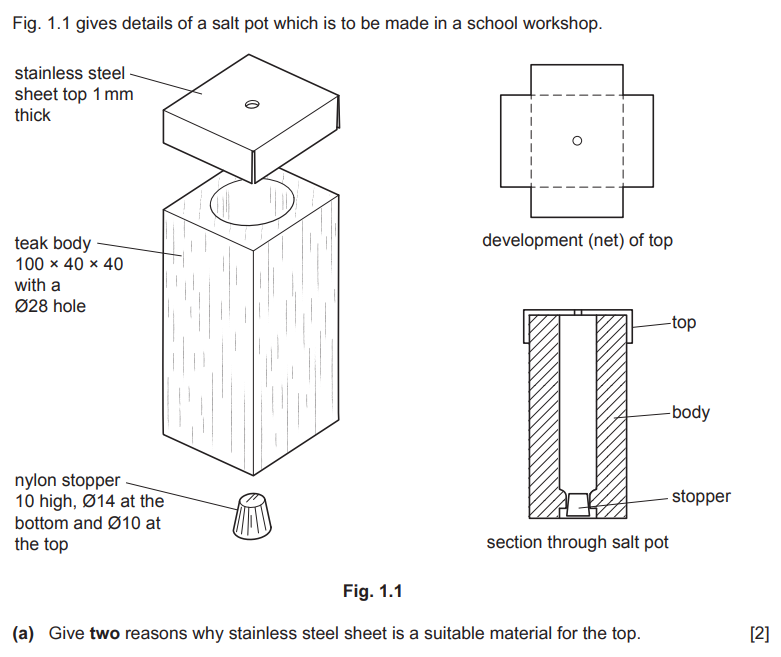

Examiner comment: Generally, this was answered well and most candidates could give at least one reason why stainless steel was suitable.
A2 Design & Technology examination questions



Note: This was originally an AS Design & Technology question, however case hardening is now part of the A2 syllabus.

Examiner comment: Candidates that knew this process answered with good detail communicating their knowledge very well.
